Genetic Roots of SMA: Unraveling Causes, Types, and Emerging Treatments
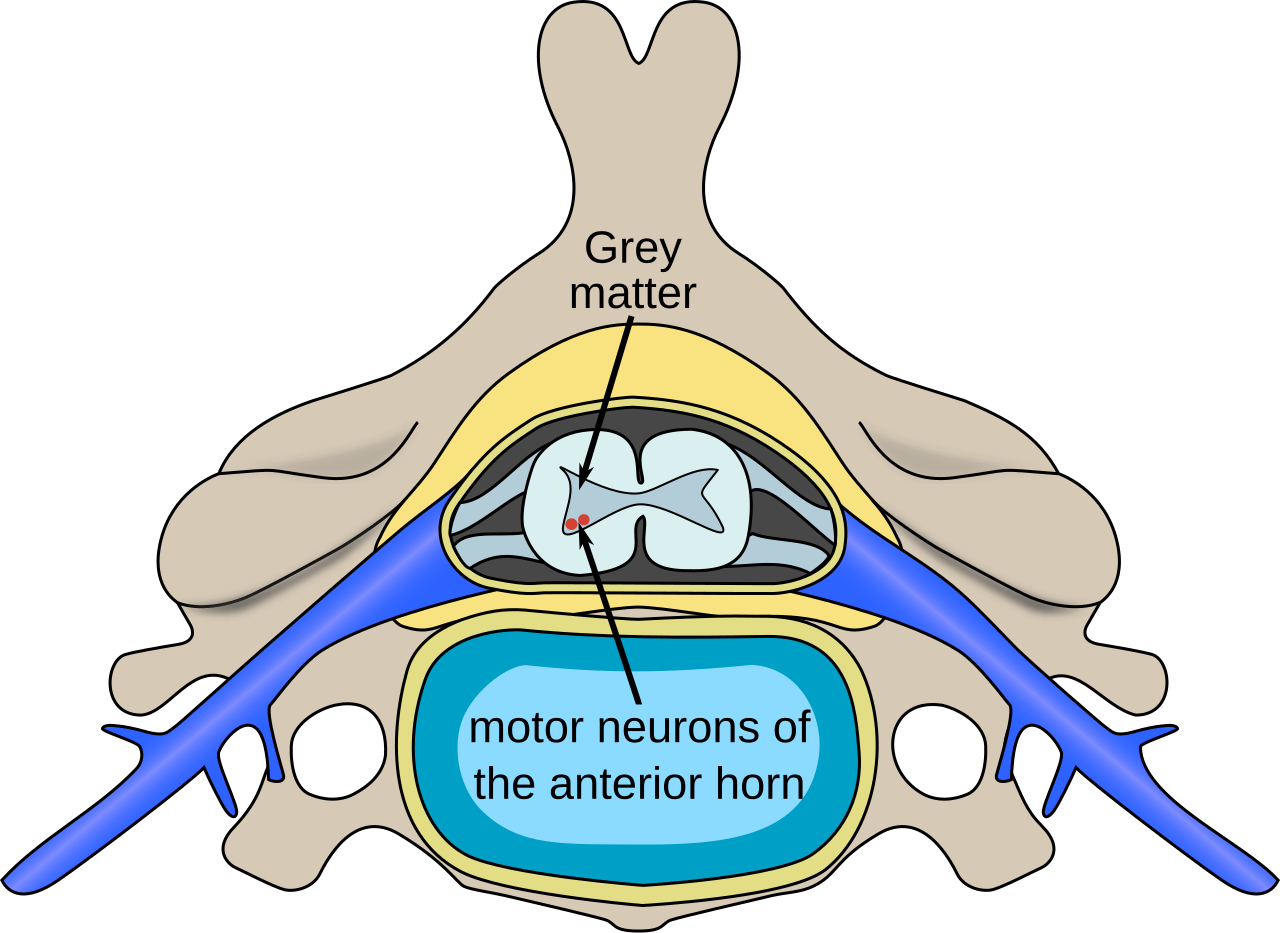
Spinal Muscular Atrophy (SMA) is a complex and challenging *neuromuscular Erkrankung* (disease) that profoundly impacts motor function. Often mistaken or broadly categorized as an "Smr Erkrankung" due to its phonetic similarity or a general search for rare muscular disorders, it's crucial to understand that SMA specifically refers to a group of genetic conditions characterized by the progressive degeneration of motor neurons. These vital nerve cells, located in the spinal cord and brainstem, are responsible for controlling voluntary muscle movement. When they falter, the brain's signals can no longer reach the muscles effectively, leading to muscle weakness, atrophy, and significant functional impairment.
SMA is a relatively rare condition, making precise prevalence data challenging to ascertain. Estimates suggest it affects between 1 in 25,000 to 1 in 75,000 live births globally. In Germany alone, it is believed that around 1,000 to 1,500 individuals live with SMA. The rarity of an "Smr Erkrankung" like SMA underscores the importance of increased awareness, robust diagnostic protocols, and access to advanced treatments to improve patient outcomes. This article delves into the fundamental genetic causes of SMA, explores its various types, and sheds light on the groundbreaking treatments that are transforming the lives of those affected.
The Genetic Blueprint: Understanding the Roots of SMA
At its core, Spinal Muscular Atrophy is a genetic disorder, meaning it's caused by a fault in an individual's DNA. The primary culprit behind SMA is a deficiency in the *Survival Motor Neuron 1 (SMN1)* gene. Normally, this gene produces a critical protein called SMN protein, which is essential for the healthy function and survival of motor neurons. In individuals with SMA, the *SMN1* gene is either missing or mutated, leading to an insufficient production of this vital protein. Without adequate SMN protein, motor neurons begin to degenerate and die, resulting in the progressive muscle weakness characteristic of the disease.
However, the story doesn't end with *SMN1*. Humans also possess a "backup" gene known as *SMN2*. While *SMN2* is very similar to *SMN1*, it typically produces a shorter, less functional version of the SMN protein. Crucially, the number of *SMN2* gene copies an individual has plays a significant role in determining the severity of their SMA. Generally, the more *SMN2* copies present, the more functional SMN protein can be produced, leading to a milder disease course. This genetic nuance explains the wide spectrum of SMA types and severities. Understanding this intricate genetic interplay is vital for accurate diagnosis and for developing targeted therapies. For more detailed insights into the genetic mechanisms, refer to our article on
SMA Disease Explained: Symptoms, Genetics, and Treatment Options.
The Spectrum of SMA: Different Types and Their Manifestations
SMA is not a monolithic condition; it presents across a wide spectrum of severity, traditionally categorized into several types based on the age of onset and the highest motor milestone achieved. These classifications help clinicians understand the likely progression and guide treatment strategies for each individual facing this "Smr Erkrankung."
*
SMA Type 1 (Severe Infantile Onset): Also known as Werdnig-Hoffmann disease, this is the most common and most severe form, with symptoms appearing within the first six months of life, often shortly after birth. Infants with Type 1 SMA experience profound muscle weakness, difficulty breathing and swallowing, and an inability to achieve basic motor milestones like head control or sitting independently. Untreated, the progressive weakness of respiratory muscles often leads to life-threatening complications, with most children not surviving beyond their second birthday. Early diagnosis, often via newborn screening, is absolutely critical for this type.
*
SMA Type 2 (Intermediate Onset): Symptoms typically emerge between 6 and 18 months of age. Children with Type 2 SMA learn to sit independently but usually do not achieve the ability to stand or walk without support. While they generally survive into adulthood, they often face significant challenges with mobility, require assistive devices, and may develop complications such as scoliosis (spinal curvature) and joint contractures. Respiratory and swallowing difficulties can develop later in the disease course, impacting quality of life.
*
SMA Type 3 (Juvenile Onset): Also known as Kugelberg-Welander disease, this milder form manifests after 18 months of age, sometimes even in adolescence or early adulthood. Individuals with Type 3 SMA achieve the ability to walk independently at some point in their lives, though they may experience a progressive decline in motor function, eventually requiring mobility aids or losing the ability to walk entirely. Respiratory and swallowing muscles are rarely severely affected, and life expectancy is typically normal, even without treatment.
*
SMA Type 4 (Adult Onset): This is the rarest and mildest form, with symptoms appearing in adulthood. It causes mild to moderate muscle weakness, typically affecting the proximal muscles (shoulders, hips) first. It progresses slowly, and individuals usually maintain their ability to walk and have a normal life expectancy.
The variability across these types highlights the importance of individualized care plans.
Diagnosis and the Lifesaving Role of Early Detection
Diagnosing SMA has historically involved a combination of clinical observation and neurological examination, followed by confirmatory tests. Today, the definitive diagnosis of SMA is made through a genetic test performed on a blood sample. This test identifies the absence or mutation of the *SMN1* gene and determines the number of *SMN2* gene copies, providing crucial information about the disease type and potential severity.
A monumental advancement in SMA management is the advent of newborn screening programs. Recognizing the critical window for intervention, many countries, including Germany, are increasingly implementing routine newborn screening for SMA. This enables the earliest possible diagnosis, often before symptoms even appear. For Type 1 SMA, where every day counts, early diagnosis means initiating treatment as soon as possible, dramatically improving outcomes and often preventing the most severe consequences of the disease. This proactive approach to identifying an "Smr Erkrankung" like SMA in its infancy is a game-changer, offering a chance at a life previously unimaginable for many affected infants. Further information on this topic can be found in
Spinale Muskelatrophie: Understanding Symptoms & Early Diagnosis.
A New Horizon: Emerging Treatments and Comprehensive Care
For decades, the management of SMA was primarily supportive, focusing on physical therapy, respiratory support, and nutritional care to mitigate symptoms and improve quality of life. While these non-medicinal therapies remain critical components of comprehensive care, the landscape of SMA treatment has been revolutionized by groundbreaking medical advancements in recent years.
We are now in an era where effective treatments can significantly alter the disease course, offering hope where there was once despair. These emerging therapies largely fall into two categories:
1.
Gene Therapy: This revolutionary approach aims to deliver a functional copy of the *SMN1* gene to motor neuron cells, enabling them to produce the essential SMN protein. Typically administered as a single intravenous infusion, gene therapy holds the promise of a long-term, if not permanent, solution, particularly when given early in life.
2.
SMN2-Splicing Modifiers: These medications work by instructing the *SMN2* gene to produce more full-length, functional SMN protein. By enhancing the natural "backup" gene, these treatments increase the overall SMN protein levels, thereby supporting motor neuron survival and improving muscle function. These are often administered regularly, either via intrathecal injection (into the spinal fluid) or orally.
These medical breakthroughs have transformed the prognosis for individuals with SMA, particularly when treatment is initiated pre-symptomatically or very early in the disease course. Children who would historically succumb to Type 1 SMA can now survive, breathe independently, and even achieve motor milestones previously thought impossible.
However, comprehensive care for any "Smr Erkrankung" like SMA extends beyond medication. A multidisciplinary team approach is essential, involving neurologists, pulmonologists, gastroenterologists, physical therapists, occupational therapists, speech and language pathologists, and genetic counselors. This team works together to manage symptoms, prevent complications (such as scoliosis or respiratory infections), support development, and enhance overall quality of life throughout the patient's lifespan. Nutritional support, respiratory management, and physical rehabilitation are ongoing pillars of care that complement the effects of disease-modifying therapies.
Conclusion
The journey to understand and treat Spinal Muscular Atrophy has seen remarkable progress, moving from a diagnosis with limited options to one with genuine hope for significantly improved outcomes. The profound understanding of SMA's genetic roots – specifically the roles of the *SMN1* and *SMN2* genes – has paved the way for innovative therapies that target the underlying cause of the disease. While the term "Smr Erkrankung" might broadly refer to a range of conditions, it is crucial to recognize SMA as a distinct, genetically defined disorder now benefiting from advanced science. With the advent of newborn screening and powerful new treatments, the focus has shifted dramatically: from managing an inevitable decline to actively preventing symptoms and enabling individuals with SMA to achieve greater independence and a higher quality of life. The ongoing commitment to research, early diagnosis, and comprehensive, multidisciplinary care continues to light the path forward for all those affected by this challenging but increasingly treatable condition.